Loading INT detail
World Fuel Services Corporation engages in the distribution of fuel and related products and services in the aviation, marine and land transportation industries worldwide.
Its Aviation segment supplies fuel and related products and services to commercial airlines, second and third tier airlines, cargo carriers, regional and low-cost carriers, airports, fixed based operators, corporate fleets, fractional operators, private aircraft, the U.S., foreign governments, intergovernmental organizations, and military customers. This segment also offers fuel management, price risk management, ground handling, dispatch services and trip planning services, such as flight planning and scheduling, weather reports and overflight permits. Its Land segment offers fuel, heating oil, propane, natural gas, lubricants and related products and services to petroleum distributors and retail petroleum operators, as well as industrial, commercial, residential and government customers. This segment also offers management services for procuring fuel and price risk management; advisory and fulfillment solutions related to power, natural gas and other energy products; consulting, renewable fuel products, carbon management and renewable energy solutions and card payment solutions; and transaction management and commercial payment programs. Its Marine segment markets fuel, lubricants and related products and services to international container and tanker fleets, commercial cruise lines, yachts and time charter operators, offshore rig owners and operators, the U.S., foreign governments, and other fuel suppliers. Its marine fuel-related services include management services to procure fuel, cost control, quality control, and claims management services. This segment also engages in the fueling of vessels, transportation and delivery of fuel and fuel-related products.
World Fuel Services Corporation was incorporated in 1984 and is headquartered in Miami, Florida.
Chart data unavailable

Poster PS5-08-16 Reports Early Observations from the INVINCIBLE-4 Study, an Ongoing Randomized, Presurgical Phase 2 Clinical Trial for Triple Negative Breast Cancer continue to show favorable safety 50% fewer grade 3 or higher Adverse Events were observed in the INT230-6 cohort compared to the Standard of Care ("SOC") neoadjuvant chemotherapy alone cohort. Poster PS4-10-15 Describes An Overview of a Potential Phase 3 Clinical Study Design with INT230-6 plus standard of care with and without the toxic anthracycline, doxorubicin SHELTON, Conn.
The paper features a comprehensive evaluation of data, including disease control rate, overall survival, immune activation, abscopal effects, tumor necrosis, dose ranging, and safety The manuscript is open access The Company will host a webinar with the paper's lead and senior authors from the University of Southern California to discuss the results on Friday, October 31, 2025, at 9:00 AM (see below) SHELTON, Conn. , Oct. 30, 2025 /PRNewswire/ -- Intensity Therapeutics, Inc. (Nasdaq: INTS) ("Intensity" or "the Company"), a late-stage clinical biotechnology company focused on the discovery and development of proprietary cancer therapies using its non-covalent, drug-conjugation technology that creates drug products designed to kill tumors and increase immune system recognition of cancers, announces that eBioMedicine, a Lancet Discovery Science journal, has published the Company's phase 1/2 IT-01 clinical study manuscript for the treatment of metastatic or refractory cancers.
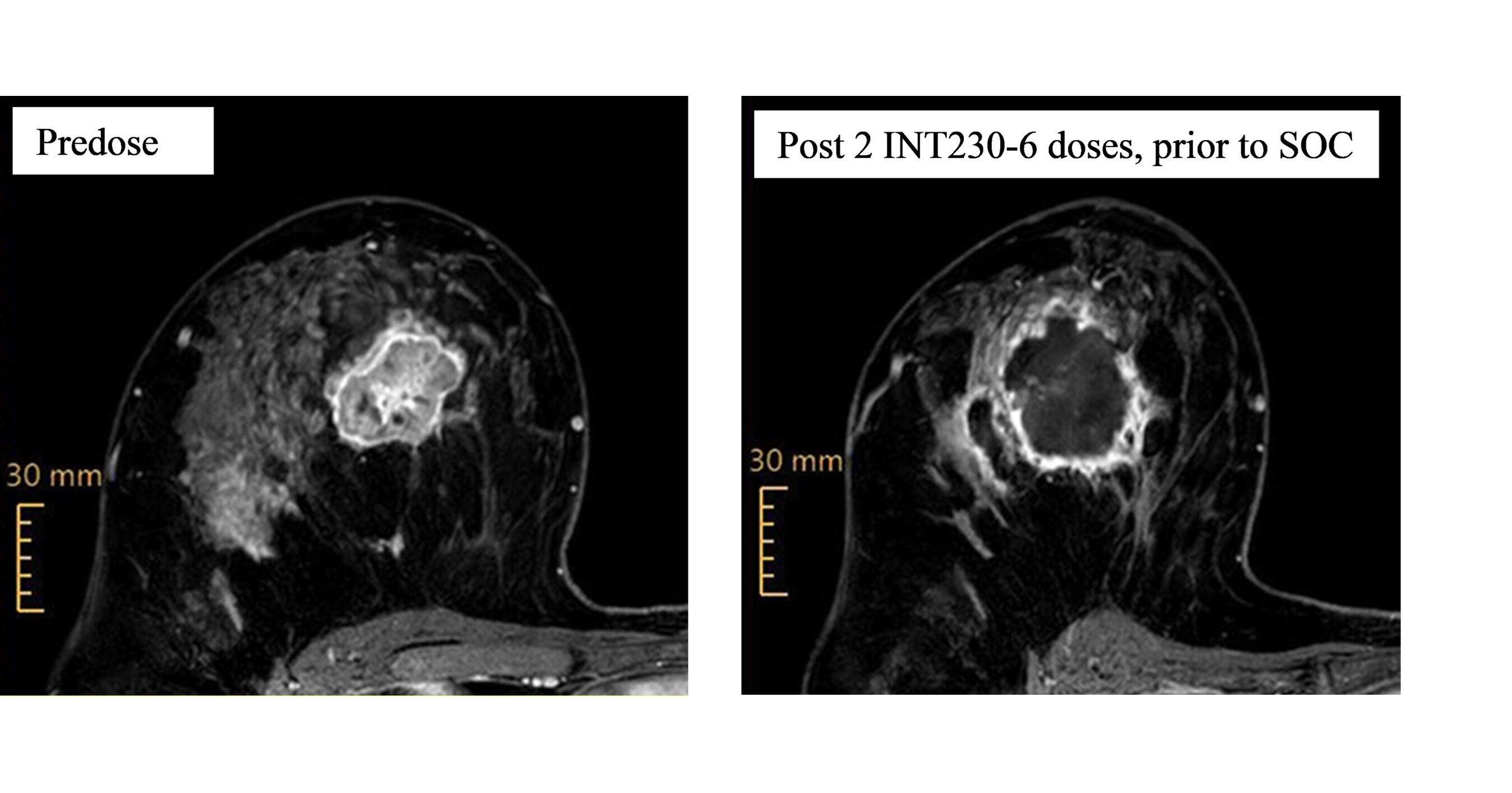
A pathological complete response ("pCR") has been observed in the first patient evaluated in Cohort A, where each patient receives two doses of INT230-6 eight days apart, followed by the standard of care immunochemotherapy ("SOC") Safety looks favorable in Cohort A Some INT230-6 patients begin to show localized skin irritation, and new patient enrollment has been paused to implement modifications to resolve the issue Patients being treated with INT230-6 continue to show significant necrosis after two doses of INT230-6 and prior to the initiation of the SOC, as shown in the photos below of a single patient's breast tumor scans SHELTON, Conn. , Sept. 10, 2025 /PRNewswire/ -- Intensity Therapeutics, Inc. ("Intensity" or "the Company") (Nasdaq: INTS), a late-stage clinical biotechnology company focused on the discovery and development of novel intratumoral cancer therapies that are designed to kill tumors and increase immune system recognition of cancers using its proprietary non-covalent conjugation technology, today provided an update on the INVINCIBLE-4 trial.

SHELTON, Conn. , June 30, 2025 /PRNewswire/ -- Intensity Therapeutics, Inc. (Nasdaq: INTS) ("Intensity" or "the Company"), a late-stage clinical biotechnology company focused on the discovery and development of proprietary, novel immune-based intratumoral cancer therapies designed to kill tumors and increase immune system recognition of cancers, announces that INT230-6 achieved complete responses in a murine models of Malignant Peripheral Nerve Sheath Tumor ("MPNST").

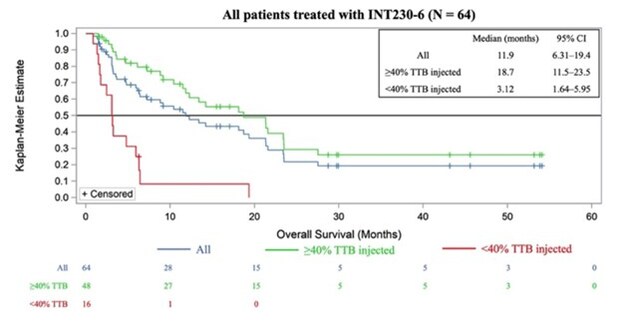
SHELTON, Conn. , June 11, 2025 /PRNewswire/ -- Intensity Therapeutics, Inc. (Nasdaq: INTS) ("Intensity" or the "Company"), a late-stage clinical biotechnology company focused on the discovery and development of proprietary, novel immune-based intratumoral cancer therapies designed to kill tumors and increase immune system recognition of cancers, announces first few patients receiving INT230-6 achieved high levels of necrosis after 8 days in the Phase 2, INVINCIBLE-4 study, before they initiated the standard-of-care regimen as shown in Figure 1.

INVINCIBLE-4 (SAKK/66/22) Study continues to recruit patients in eight sites in Switzerland SHELTON, Conn. and BERN, Switzerland , May 6, 2025 /PRNewswire/ -- Intensity Therapeutics, Inc. (Nasdaq: INTS), ("Intensity" or "the Company") a late-stage clinical biotechnology company focused on the discovery and development of proprietary, novel immune-based intratumoral cancer therapies designed to kill tumors and increase immune system recognition of cancers, and The Swiss Group for Clinical Cancer Research SAKK ("SAKK"), a decentralized academic research institute that has been conducting clinical trials of cancer treatments in all major Swiss hospitals since 1965, announced that the European Medicines Agency ("EMA") has authorized the initiation of the INVINCIBLE-4 (SAKK 66/22) ("INVINCIBLE-4 Study") (NCT06358573) in France in collaboration with Unicancer.

The US FDA, Health Canada, European Medicines Authority, and the Australian Therapeutic Goods Administration authorized the Company's global, randomized Phase 3 study (INVINCIBLE-3) in Metastatic Soft Tissue Sarcoma Twenty-three sites are currently contracted in the INVINCIBLE 3 study, and several sites have treated patients Seven Swiss sites are activated in the Phase 2 (INVINCIBLE-4) study for early-stage, operable Triple Negative Breast Cancer ("TNBC"), and several patients have been treated SHELTON, Conn. , Jan. 10, 2025 /PRNewswire/ -- Intensity Therapeutics, Inc. ("Intensity" or the "Company") (Nasdaq: INTS), a late-stage clinical biotechnology company focused on the discovery and development of proprietary, novel immune-based intratumoral cancer therapies designed to kill tumors and increase immune system recognition of cancers, today announces a business update highlighting key achievements with its lead drug candidate INT230-6.

MIAMI--(BUSINESS WIRE)--World Kinect Corporation (NYSE:WKC) (the “Company”) announced today that its board of directors has declared a quarterly cash dividend of $0.14 per share, which is payable on July 10, 2023 to shareholders of record on June 26, 2023. Earlier today, the Company announced that it had changed its name from World Fuel Services Corporation to World Kinect Corporation. The Company will begin trading under its new name and new ticker symbol (NYSE: WKC) effective tomorrow, on Jun.

MIAMI--(BUSINESS WIRE)--World Fuel Services Corporation (NYSE:INT) (“Company”) today announced it has changed its corporate name to World Kinect Corporation. The Company's shareholders approved the name change during today's Annual Meeting. The Company will begin trading under its new name and ticker symbol (NYSE: WKC) effective tomorrow, June 16, 2023. This change is intended to better reflect the Company's ongoing transformation into a more resilient, diversified energy and solutions provider.

It may be time to sell some of the top energy stocks, especially as they become overvalued. Sure, according to the International Energy Agency, rising post-Covid demand from China, coupled with tight supply, suggests that a crude oil rebound is possible later this year.

Is World Fuel Services (INT) a great pick from the value investor's perspective right now? Read on to know more.

World Fuel Services Corporation (INT) Q1 2023 Earnings Call Transcript.

World Fuel Services (INT) came out with quarterly earnings of $0.36 per share, beating the Zacks Consensus Estimate of $0.35 per share. This compares to earnings of $0.42 per share a year ago.

MIAMI--(BUSINESS WIRE)--World Fuel Services Corporation (NYSE: INT) today reported financial results for the first quarter of 2023. Results compared to the same period last year are as follows (unaudited - in millions, except percentages and per share data): Three Months Ended March 31, 2023 2022 Change Volume (1) 4,472 4,476 0 % Revenue $ 12,482 $ 12,382 1 % Gross profit $ 263 $ 231 14 % Income from operations $ 65 $ 41 56 % Income from o.

MIAMI--(BUSINESS WIRE)--World Fuel Services Corporation (NYSE:INT) invites you to participate in a conference call with its management team on Thursday, April 27, 2023 at 5:00 p.m. Eastern Time to discuss the Company's first quarter results, as well as certain forward-looking information. The Company plans to release its first quarter results after the market closes on the same date.